What Is The Molar Mass Of H2o2
sonusaeterna
Nov 29, 2025 · 11 min read
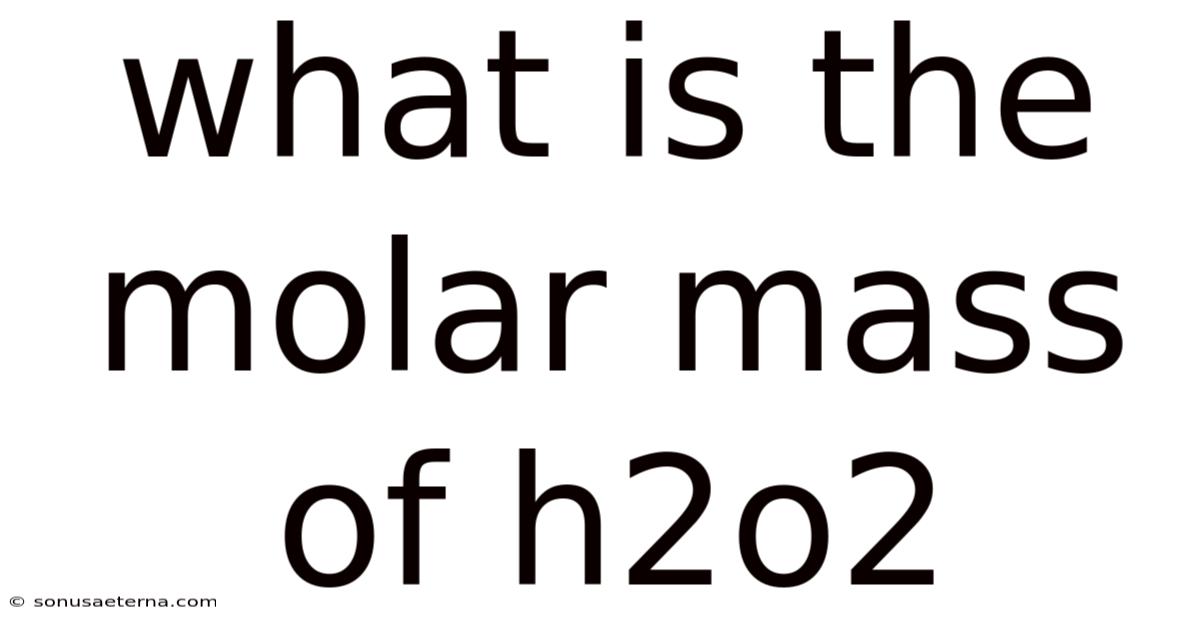
Table of Contents
Imagine needing to mix a cleaning solution for a delicate antique, or precisely calibrating a disinfectant for a hospital environment. In both scenarios, accuracy is paramount, and understanding the molar mass of hydrogen peroxide (H₂O₂) becomes crucial. This seemingly simple calculation underpins a wide range of applications, ensuring safety and effectiveness in everything from household cleaning to industrial processes.
The concept of molar mass can initially seem abstract, but it's essentially the weight of one mole of a substance – a specific number of molecules (6.022 x 10²³) that allows us to bridge the gap between the microscopic world of atoms and molecules and the macroscopic world we experience. When dealing with chemicals like hydrogen peroxide, knowing its molar mass is not just an academic exercise; it's a practical necessity for accurate measurements, dilutions, and chemical reactions. Let's delve into the details of how to calculate the molar mass of H₂O₂, its significance, and its diverse applications.
Understanding the Molar Mass of H₂O₂
The molar mass of a compound is the sum of the atomic masses of all the atoms in its chemical formula, expressed in grams per mole (g/mol). For hydrogen peroxide (H₂O₂), this means we need to consider the atomic masses of hydrogen (H) and oxygen (O) and how many of each are present in a single molecule of H₂O₂.
Defining Molar Mass
Molar mass is a fundamental concept in chemistry, linking the mass of a substance to the amount of substance in moles. One mole is defined as the amount of a substance that contains as many elementary entities (atoms, molecules, ions, etc.) as there are atoms in 12 grams of carbon-12. This number, known as Avogadro's number (approximately 6.022 x 10²³), provides a bridge between the atomic mass unit (amu) used for individual atoms and the gram, a unit we can measure in the lab. The molar mass is numerically equivalent to the atomic or molecular weight of a substance but is expressed in g/mol rather than amu.
The Scientific Foundation
The calculation of molar mass is based on the atomic masses of the elements, which are experimentally determined and listed on the periodic table. These atomic masses are relative values, with carbon-12 serving as the standard. For example, the atomic mass of hydrogen is approximately 1.008 amu, and the atomic mass of oxygen is approximately 16.00 amu. These values are crucial for determining the molar mass of any compound containing these elements. The concept of molar mass is tied to stoichiometry, the branch of chemistry that deals with the quantitative relationships between reactants and products in chemical reactions. Knowing the molar mass of each reactant and product allows chemists to accurately predict the amounts of substances needed or produced in a reaction, ensuring efficiency and minimizing waste.
A Brief History
The concept of molar mass evolved from the work of early chemists who sought to understand the relationships between the masses of elements and compounds. John Dalton's atomic theory, proposed in the early 19th century, laid the groundwork by suggesting that elements are composed of discrete atoms with characteristic masses. Later, Amedeo Avogadro proposed that equal volumes of gases, at the same temperature and pressure, contain the same number of molecules. This hypothesis eventually led to the definition of Avogadro's number and the concept of the mole. The development of accurate methods for determining atomic masses, such as mass spectrometry, further refined our understanding of molar mass and its applications.
Essential Concepts Related to Molar Mass
Several essential concepts are closely related to molar mass, including molecular weight, formula weight, and mole fraction. Molecular weight is the sum of the atomic weights of the atoms in a molecule and is a dimensionless quantity. Formula weight is used for ionic compounds and is calculated similarly to molecular weight, using the atomic weights of the ions in the formula unit. Mole fraction is the ratio of the number of moles of a particular component in a mixture to the total number of moles of all components. These concepts are all interconnected and play a crucial role in quantitative chemical analysis.
Calculating the Molar Mass of H₂O₂
To calculate the molar mass of H₂O₂, we need to add up the atomic masses of all the atoms in the molecule:
- Two hydrogen atoms (2 x atomic mass of H)
- Two oxygen atoms (2 x atomic mass of O)
Using the approximate atomic masses of H (1.008 g/mol) and O (16.00 g/mol), the calculation is as follows:
(2 x 1.008 g/mol) + (2 x 16.00 g/mol) = 2.016 g/mol + 32.00 g/mol = 34.016 g/mol
Therefore, the molar mass of H₂O₂ is approximately 34.016 g/mol. This value is essential for converting between mass and moles of hydrogen peroxide in chemical calculations.
Trends and Latest Developments
The understanding and application of molar mass remain fundamental in chemistry, but several trends and developments are worth noting. These include advancements in analytical techniques, the use of computational methods, and the increasing importance of accurate molar mass determination in fields like pharmaceuticals and materials science.
Current Trends
One significant trend is the increasing use of sophisticated analytical techniques, such as mass spectrometry and chromatography, for accurate determination of molar masses. These techniques allow scientists to identify and quantify even trace amounts of substances, providing valuable information for research and quality control. Another trend is the use of computational methods, such as density functional theory (DFT), to predict and verify molar masses. These methods can be particularly useful for complex molecules or materials where experimental data is limited.
Data and Popular Opinions
There is a growing awareness of the importance of accurate molar mass determination in various fields. In pharmaceuticals, for example, the correct molar mass is crucial for calculating the appropriate dosage of a drug. In materials science, it is essential for characterizing the composition and properties of new materials. Popular opinion among chemists and researchers is that continued advancements in analytical and computational techniques will further improve our ability to determine and utilize molar mass information.
Professional Insights
From a professional standpoint, the ability to accurately determine and apply molar mass is a critical skill for chemists and related professionals. It is essential for conducting research, developing new products, and ensuring the safety and quality of chemical processes. Professionals in fields such as pharmaceuticals, environmental science, and materials science rely on molar mass calculations daily to perform their work effectively. Staying up-to-date with the latest advancements in analytical techniques and computational methods is crucial for maintaining expertise in this area.
Tips and Expert Advice
Working with molar mass calculations can be straightforward, but attention to detail is essential to ensure accuracy. Here are some practical tips and expert advice to help you master the use of molar mass in various applications.
Double-Check Atomic Masses
Always verify the atomic masses you are using from a reliable source, such as the periodic table published by IUPAC (International Union of Pure and Applied Chemistry). Atomic masses can vary slightly depending on the isotope ratios of the element, so using the most up-to-date and accurate values is essential. Pay attention to the number of significant figures in the atomic masses and use the appropriate number of significant figures in your final answer to avoid rounding errors.
For example, if you are using the atomic mass of hydrogen as 1.008 g/mol and the atomic mass of oxygen as 16.00 g/mol, your final answer for the molar mass of H₂O₂ should have at least four significant figures. This level of precision is often necessary for accurate chemical calculations and measurements.
Understand the Context
Consider the context in which you are using molar mass. Are you calculating the amount of reactant needed for a chemical reaction, determining the concentration of a solution, or characterizing a new material? Understanding the context will help you choose the appropriate formula and method for your calculation. Be aware of any potential sources of error, such as impurities in your sample or limitations in your analytical techniques.
For instance, if you are using hydrogen peroxide as a disinfectant, you need to know the exact concentration of the solution to ensure that it is effective against the target microorganisms. In this case, you would need to use the molar mass of H₂O₂ to convert between mass and moles and then calculate the concentration in terms of molarity (moles per liter).
Use Dimensional Analysis
Dimensional analysis, also known as the factor-label method, is a powerful tool for ensuring that your calculations are set up correctly. This method involves tracking the units of each quantity and making sure that they cancel out to give you the desired units in your final answer. For example, if you are converting from grams to moles, you would multiply by the reciprocal of the molar mass (i.e., moles per gram).
To illustrate, suppose you have 50 grams of H₂O₂ and you want to know how many moles this represents. You would set up the calculation as follows:
50 g H₂O₂ x (1 mol H₂O₂ / 34.016 g H₂O₂) = 1.47 mol H₂O₂
Notice how the units of grams cancel out, leaving you with the desired units of moles.
Practice Regularly
Like any skill, mastering molar mass calculations requires practice. Work through various examples and problems to build your confidence and understanding. Start with simple calculations and gradually move on to more complex ones. Use online resources, textbooks, and practice problems to reinforce your learning. Don't be afraid to ask for help from teachers, tutors, or fellow students if you are struggling with a particular concept.
For example, try calculating the molar mass of other common compounds, such as water (H₂O), sodium chloride (NaCl), and glucose (C₆H₁₂O₆). Then, try using these molar masses to solve stoichiometry problems, such as calculating the amount of product formed in a chemical reaction.
Stay Updated
Keep abreast of the latest developments in chemistry and related fields. New techniques and technologies are constantly being developed that can improve the accuracy and efficiency of molar mass determination. Attend conferences, read scientific journals, and participate in online forums to stay informed about these advancements. Consider taking continuing education courses or workshops to update your skills and knowledge.
For instance, advancements in mass spectrometry have made it possible to determine the molar masses of even very large and complex molecules, such as proteins and polymers. These techniques are invaluable for researchers in fields like biochemistry and materials science.
FAQ
Q: What is the difference between molar mass and molecular weight? A: Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol), while molecular weight is the sum of the atomic weights of the atoms in a molecule and is a dimensionless quantity. Numerically, they are very similar, but molar mass includes the units.
Q: How does the molar mass of an element relate to its atomic mass? A: The molar mass of an element is numerically equal to its atomic mass, but the units are different. The atomic mass is expressed in atomic mass units (amu), while the molar mass is expressed in grams per mole (g/mol).
Q: Why is it important to know the molar mass of a substance? A: Knowing the molar mass of a substance is essential for converting between mass and moles, which is crucial for performing accurate chemical calculations, preparing solutions, and conducting chemical reactions.
Q: Can the molar mass of a compound be determined experimentally? A: Yes, the molar mass of a compound can be determined experimentally using techniques such as mass spectrometry, which measures the mass-to-charge ratio of ions.
Q: How does the molar mass of an isotope affect the molar mass of a compound? A: The molar mass of an isotope affects the average atomic mass of an element, which in turn affects the molar mass of a compound. The natural abundance of isotopes must be considered when calculating the molar mass of a compound.
Conclusion
Understanding the molar mass of hydrogen peroxide (H₂O₂) is essential for accuracy in various applications, from household cleaning to industrial processes. By summing the atomic masses of its constituent atoms, we find that the molar mass of H₂O₂ is approximately 34.016 g/mol. This knowledge enables precise calculations for dilutions, reactions, and the safe handling of this versatile chemical.
Now that you have a comprehensive understanding of the molar mass of H₂O₂, put your knowledge into practice! Try calculating the molar masses of other common compounds, or explore the use of H₂O₂ in different applications. Share your findings and questions in the comments below, and let's continue to explore the fascinating world of chemistry together.
Latest Posts
Latest Posts
-
Why Is Measurement Important In Science
Nov 29, 2025
-
Who Painted Adoration Of The Magi
Nov 29, 2025
-
How Do You Say Alright In Spanish
Nov 29, 2025
-
How Big Was A Viking Longship
Nov 29, 2025
-
What Are Some Fun Facts About The Tundra
Nov 29, 2025
Related Post
Thank you for visiting our website which covers about What Is The Molar Mass Of H2o2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.